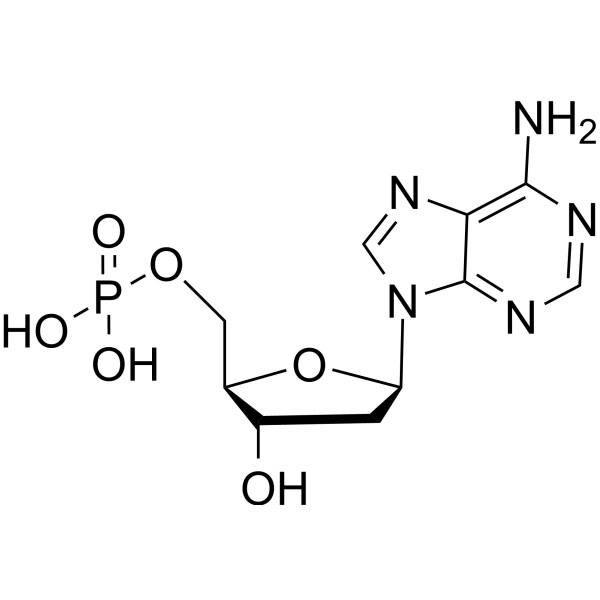
2′-Deoxyadenosine-5′-Monophosphate Free Acid (dAMP) CAS 653-63-4 HPLC Purity ≥98.0%
Manufacturer Supply Deoxynucleotide Intermediates with High Purity and Stable Quality
2'-Deoxyadenosine-5'-Monophosphate Disodium Salt (dGMP.Na2) CAS: 33430-61-4
2'-Deoxyadenosine-5'-Monophosphate Free Acid (dAMP) CAS: 653-63-4
2'-Deoxycytidine-5'-Monophosphate Hydrate (dCMP) CAS: 1032-65-1
| Chemical Name | 2'-Deoxyadenosine-5'-Monophosphate Free Acid |
| Synonyms | dAMP; 5'-dAMP; 2'-Deoxyadenosine 5'-Phosphate |
| CAS Number | 653-63-4 |
| Stock Status | In Stock |
| Molecular Formula | C10H14N5O6P |
| Molecular Weight | 331.22 |
| Melting Point | 148℃ |
| Solubility | Soluble in 1N Ammonium Hydroxide (50 mg/ml) and Water |
| COA & MSDS | Available |
| Place of Origin | Shanghai, China |
| Brand | Ruifu Chemical |
| Item | Specifications | Specifications |
| Appearance | White Crystalline Powder | White Crystalline Powder |
| Transmission | ≥95.0% | 97.2% |
| Assay | ≥90.0% (UV Spectrophotom) | 98.5% |
| A250/A260 | 0.79~0.85 | 0.82 |
| A280/A260 | 0.20~0.26 | 0.24 |
| Water Content | ≤10.0% | 5.5% |
| Heavy Metals | ≤10ppm | <10ppm |
| Arsenic (As) | ≤2ppm | <2ppm |
| Purity / Analysis Method | ≥98.0% (HPLC) | 98.6% (HPLC) |
| pH Value | 2.0~3.5 | 3.1 |
| Test Standard | Enterprise Standard | Conforms |
Package: Bottle, Aluminum foil bag, Cardboard drum, 25kg/Drum, or according to customer's requirement.
Storage Condition: Store in sealed containers at cool and dry place; Protect from sunlight, moisture

Risk Codes
R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed.
R36/37/38 - Irritating to eyes, respiratory system and skin.
Safety Description
S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36 - Wear suitable protective clothing.
S37/39 - Wear suitable gloves and eye/face protection
S24/25 - Avoid contact with skin and eyes.
WGK Germany 3
HS Code 2934993090
2′-Deoxyadenosine 5′-Monophosphate (CAS 653-63-4) is used to study adenosine-based cellular interactions. 2'-Deoxyadenosine-5'-Monophosphate is a derivative of the nucleic acid, AMP, in which the hydroxyl group on the 2' carbon of the pentose has been reduced to a hydrogen atom. It has been used in the synthesis of novel photoaffinity labels for incorporation into DNA and to study adenosine-based interactions during DNA synthesis and DNA damage.
-
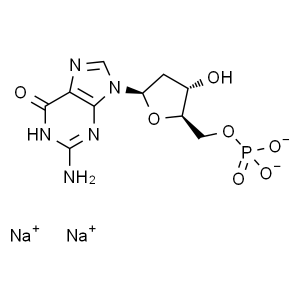
Disodium 5′-dGMP (dGMP.Na2) CAS 33430-61-...
-
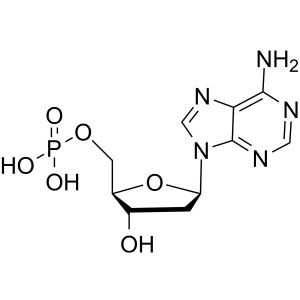
2′-Deoxyadenosine-5′-Monophosphate ...
-
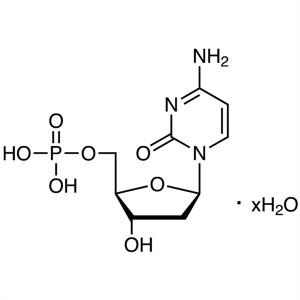
2′-Deoxycytidine 5′-Monophosphate H...
-
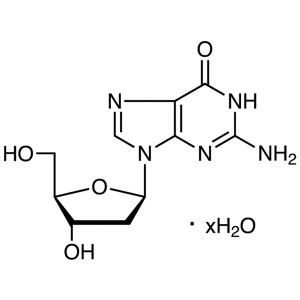
2′-Deoxyguanosine monohydrate CAS 961-07-...
-
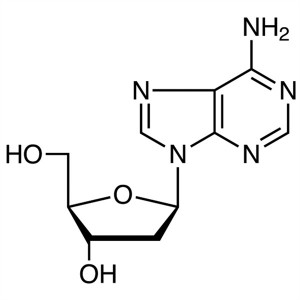
2′-Deoxyadenosine CAS 958-09-8 Purity ≥99...
-
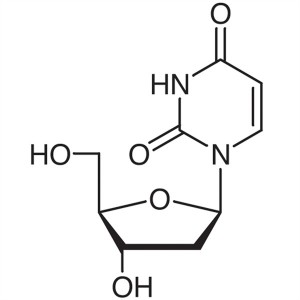
2′-Deoxyuridine CAS 951-78-0 Purity ≥99.0...
-
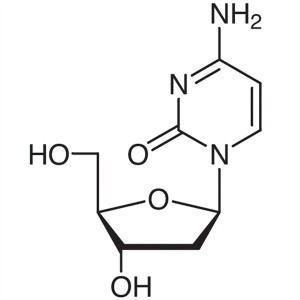
2′-Deoxycytidine CAS 951-77-9 Purity ≥99....
-
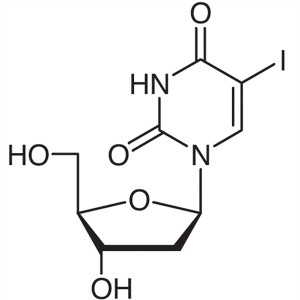
5-Iodo-2′-Deoxyuridine (5-IUdR) CAS 54-42...
-
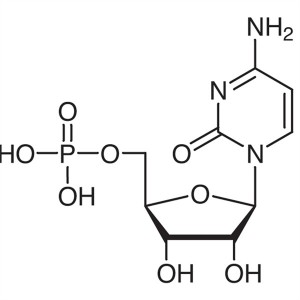
Cytidine 5′-Monophosphate (5′-CMP) ...
-
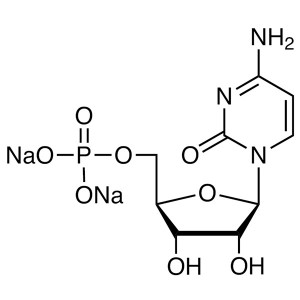
Cytidine 5′-Monophosphate Disodium Salt (...
-
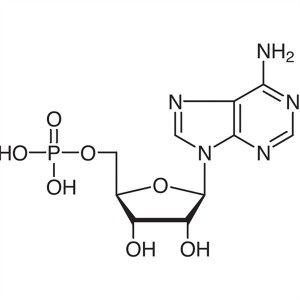
Adenosine 5′-Monophosphate (5′-AMP)...
-
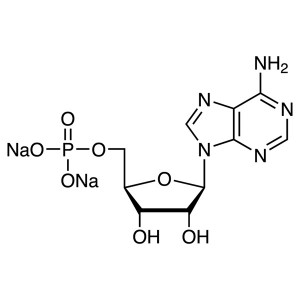
Adenosine 5′-Monophosphate Disodium Salt ...
-
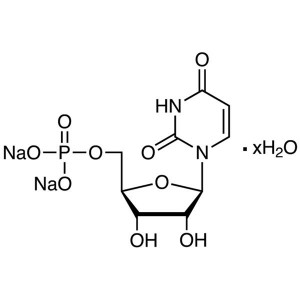
Uridine 5′-Monophosphate Disodium Salt Hy...
-
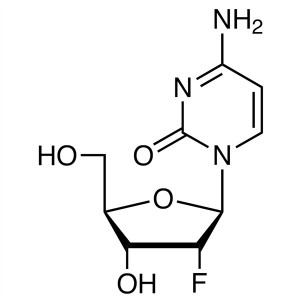
2′-Deoxy-2′-Fluorocytidine CAS 1021...
-
2′-Deoxy-2′-Fluorouridine CAS 784-7...
-
Guanosine CAS 118-00-3 Purity ≥98.0% (HPLC) Ass...